MFM Pellet contains several important amino acids, including cysteine, a key component in many antioxidants. Also included in MFM Pellet are the branched-chain amino acids (BCAA) leucine, isoleucine, and valine, all three of which are essential amino acids that must be obtained from the diet. BCAA may be used as a substrate for energy production in skeletal muscle. Research in human athletes has shown that BCAA may stimulate protein synthesis and delay onset of fatigue. MFM Pellet also contains the essential amino acids lysine, methionine, and threonine.
The understanding of muscle disease in horses has broadened considerably over the last few decades. Researchers continue to discover nuances in muscle dysfunction, and one disease continues to gain recognition within the Warmblood and endurance communities: myofibrillar myopathy (MFM).
Like those diagnosed with polysaccharide storage myopathy (PSSM), horses with MFM demonstrate exercise intolerance and occasional episodes of tying-up. Microscopic examination of muscle tissue reveals, however, that the underlying cause of MFM is much different than it is for PSSM.
Use MFM Pellet to increase the amount of key amino acids and high-quality protein to protect the muscle from oxidative stress and rebuild damaged muscle fibers.
“In our studies, horses with MFM improved considerably once their diets were modified to include the recommended ratios of energy sources and MFM Pellet to support muscle repair and recovery.”
Stephanie Valberg, D.V.M., Ph.D., Dipl. ACVIM, ACVSMR, Director of the Valberg Equine Neuromuscular Diagnostic Laboratory
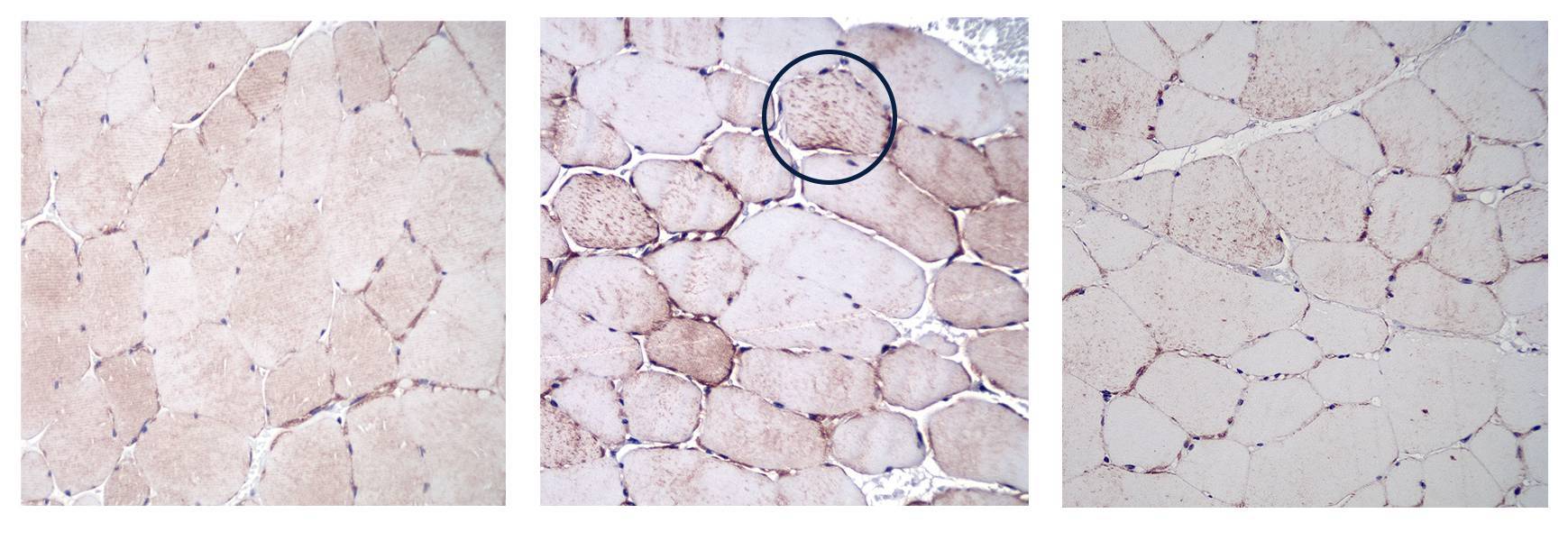 Cross sections of muscle cells. Left, a healthy horse. Center, a horse with MFM showing desmin aggregates in the muscle. Right, the same horse on MFM Pellet.
Cross sections of muscle cells. Left, a healthy horse. Center, a horse with MFM showing desmin aggregates in the muscle. Right, the same horse on MFM Pellet.
Sarcomeres are the basic contractile units of muscle fiber. Each sarcomere is composed of two main protein filaments, actin and myosin, which are the structures responsible for muscular contraction. These structures lie side by side in a staggered formation, while another protein called desmin stabilizes the filaments to maintain their alignment. In horses with MFM, the alignment between the actin and myosin strands fails, causing tears and gaps in the filaments. Muscle biopsies of affected horses show abnormal desmin deposits at these disruption sites.
While researchers remain unsure why these changes in myofibrils occur, MFM has been identified and studied most extensively in two groups of horses, Warmbloods and Arabians. Crossbred horses of Arabian and Warmblood breeding as well as horses of unrelated breeds have also been affected. Warmbloods typically show exercise intolerance by the time they are six to eight years of age. This is characterized by an unwillingness to go forward, inability to achieve or maintain collection, reduced capacity for routine work, and difficulty with upward transitions to canter. Muscle pain, stiffness, and vague hindlimb lameness are also signs of MFM in Warmbloods.
According to Valberg, the “basis for MFM in Warmblood horses appears to be related to the individual effects of diet, exercise, and training on gene and protein responses to exercise with downstream effects on muscle mass, the alignment of contractile proteins, mitochondrial function, and oxidative stress.”
Sarcomeres are the basic contractile units of muscle fiber. Each sarcomere is composed of two main protein filaments, actin and myosin, which are the structures responsible for muscular contraction. These structures lie side by side in a staggered formation, while another protein called desmin stabilizes the filaments to maintain their alignment. In horses with MFM, the alignment between the actin and myosin strands fails, causing tears and gaps in the filaments. Muscle biopsies of affected horses show abnormal desmin deposits at these disruption sites.
While researchers remain unsure why these changes in myofibrils occur, MFM has been identified and studied most extensively in two groups of horses, Warmbloods and Arabians. Crossbred horses of Arabian and Warmblood breeding as well as horses of unrelated breeds have also been affected. Warmbloods typically show exercise intolerance by the time they are six to eight years of age. This is characterized by an unwillingness to go forward, inability to achieve or maintain collection, reduced capacity for routine work, and difficulty with upward transitions to canter. Muscle pain, stiffness, and vague hindlimb lameness are also signs of MFM in Warmbloods.
Note that these signs are not specific to MFM and they can occur with poor saddle fit, lameness from hock or stifle degeneration, suspensory injuries, sacroiliac disorders, inflammation of thoracic or lumbar vertebrae, and more. A thorough evaluation for lameness is needed to rule out these more common issues and should precede any testing for MFM horses.
According to Valberg, the “basis for MFM in Warmblood horses appears to be related to the individual effects of diet, exercise, and training on gene and protein responses to exercise with downstream effects on muscle mass, the alignment of contractile proteins, mitochondrial function, and oxidative stress.”
Arabians diagnosed with MFM are typically engaged in endurance riding. Horses have irregular elevations in serum creatine kinase activity after endurance rides or during exercise that follows a week or more of rest. Elevated creatine kinase is a sign of muscle damage. Other signs of tying-up, such as sweating, pain, and reluctance to move, are hit-or-miss and oftentimes less severe than those signs exhibited by horses with other forms of exertional rhabdomyolysis. Discolored urine has been noted in Arabian horses with only slight muscle stiffness.
The basis for MFM in Arabian horses appears to be related to a need for increased cysteine synthesis, decreased cysteine-based antioxidants, and oxidative stress. With this in mind, these horses would have an increased requirement for cysteine following exercise.
Arabians diagnosed with MFM are typically engaged in endurance riding. Horses have irregular elevations in serum creatine kinase activity after endurance rides or during exercise that follows a week or more of rest. Elevated creatine kinase is a sign of muscle damage. Other signs of tying-up, such as sweating, pain, and reluctance to move, are hit-or-miss and oftentimes less severe than those signs exhibited by horses with other forms of exertional rhabdomyolysis. Discolored urine has been noted in Arabian horses with only slight muscle stiffness.
The basis for MFM in Arabian horses appears to be related to a need for increased cysteine synthesis, decreased cysteine-based antioxidants, and oxidative stress. With this in mind, these horses would have an increased requirement for cysteine following exercise.
Like PSSM2, the diagnosis can only be made by muscle biopsy and there are no valid genetic tests for MFM. Diagnosis must be made by muscle biopsy. More information about testing for MFM can be found HERE.
Research Proven
A dressage horse was presented for veterinary evaluation over a three-year period for performance-related complaints. These included exercise intolerance, reluctance to engage his hindquarters under saddle, unwillingness to go forward, and behavioral changes when ridden. After supplementation with MFM Pellet for three months, the gelding improved dramatically.
Nutritional Management
Researchers continue to delve into the intricacies of this recently discovered myopathy, including how best to nourish affected horses. Rations for horses diagnosed with MFM should focus on providing high-quality protein and specific amino acids, which may support the regeneration of sarcomeres.
Horses with MFM should consume 1.5%–2% of body weight per day of hay; use good-quality grass or mixed hays (aim for a range of 55%–65% NDF, 10%–12% CP, 10%–17% NSC). If concentrates are required to maintain body condition, look for products with moderate levels of NSC (20-30% NSC) and lower fat (4-6%), as fat increases oxidative stress. Contact us for recommended products available in your area. Use MFM Pellet to increase the amount of key amino acids and high-quality protein to protect the muscle from oxidative stress and rebuild damaged muscle fibers.
MFM Pellet Feeding Directions
Top-dress on feed. Divide between two meals. Feed 120 g per meal twice daily.
Nutritional Management
Researchers continue to delve into the intricacies of this recently discovered myopathy, including how best to nourish affected horses. Rations for horses diagnosed with MFM should focus on providing high-quality protein and specific amino acids, which may support the regeneration of sarcomeres.
Horses with MFM should consume 1.5%–2% of body weight per day of hay; use good-quality grass or mixed hays (aim for a range of 55%–65% NDF, 10%–12% CP, 10%–17% NSC). If horses have lower energy needs, feed a ration balancer pellet such as KER All-Phase®. If concentrates are required to maintain body condition, look for products with moderate levels of NSC (20-30% NSC) and lower fat (4-8%), as fat increases oxidative stress. Contact us for recommended products available in your area. Use MFM Pellet to increase the amount of key amino acids and high-quality protein to protect the muscle from oxidative stress and rebuild damaged muscle fibers.
Ingredients
MFM Pellet Feeding Directions
Top-dress on feed. Divide between two meals. Feed 120 g per meal twice daily. Feed Arabians 90-120 g twice daily.
MFM Pellet has a strong, distinct smell that is characteristic of the ingredients. Introduce it gradually over 7-14 days and use molasses or sweet feed to improve acceptance by finicky horses.
If additional antioxidant support is needed, use Nano-Q10™ to supply coenzyme Q10 and Nano-E® to supply bioavailable natural vitamin E. For more information, contact Kentucky Equine Research at 1800 772 198.
About KERx Special Needs Nutrition
KERx is a division of KER Targeted Nutrition featuring products developed by Kentucky Equine Research and recommended by veterinarians to support specific nutrition-related challenges.
References
Hauss, A., C. Loos, A. Gerritsen, K. Urschel, and J. Pagan. 2021. Effect of branched-chain amino acid and N-acetylcysteine supplementation post-exercise on muscle mTOR signaling in exercising horses. Journal of Equine Veterinary Science 100:103524.
Valberg, S.J. 2021. Diagnosis and management of myofibrillar myopathy in warmblood performance horses. Proc AAEP 67:214-218.
Pagan, J.D., Valberg, S.J. 2020. Feeding performance horses with myopathies. Proc AAEP 66:66-74.
Valberg, S.J., Finno, C.J., Henry, M.L., et al. 2020. Commercial genetic testing for type 2 polysaccharide storage myopathy and myofibrillar myopathy does not correspond to a histopathological diagnosis. Equine Vet J; 0:1-11.
Williams, Z.J., D. Velez-Irizarry, J.L. Petersen, J. Ochala, C.J. Finno, and S.J. Valberg. 2020. Candidate gene expression and coding sequence variants in Warmblood horses with myofibrillar myopathy. Equine Veterinary Journal: doi.org/10.1111/evj.13286.
Henry MI, Velez-Irizarry D, Pagan JD, et al. 2019. Impact of antioxidant supplementation on plasma thiols and the muscle proteome in Thoroughbred horses. J Equine Vet Sci; 76:84.
Valberg, S.J., Perumbakkam, S., McKenzie, E.C., et al. 2018. Proteome and transcriptome profiling of equine myofibrillar myopathy identifies diminished peroxiredoxin 6 and altered cysteine metabolic pathways. Physiol Genomics; 50:1036-1050.
Valberg, S.J., A.M. Nicholson, S.S. Lewis, R.A. Reardon, and C.J. Finno. 2017. Clinical and histopathological features of myofibrillar myopathy in Warmblood horses. Equine Veterinary Journal 49:739-745.
McKenzie, E.C., L.V. Eyrich, M.E. Payton, and S.J. Valberg. 2016. Clinical, histopathological and metabolic responses following exercise in Arabian Horses with a history of exertional rhabdomyolysis. Veterinary Journal 216:196-201.
Valberg, S.J., E.C. McKenzie, L.V. Eyrich, J. Shivers, N.E. Barnes, and C.J. Finno. 2016. Suspected myofibrillar myopathy in Arabian horses with a history of exertional rhabdomyolysis. Equine Veterinary Journal 48:548-556.








