Myofibrillar Myopathy
What is Myofibrillar Myopathy (MFM)?
Myofibrillar myopathy or MFM is a term used to describe a new disease our research group has identified in horses, particularly in Arabian and Warmblood horses. Previously, some horses with MFM have been diagnosed with type 2 PSSM because the breaks in myofibrils created gaps that were filled in by muscle glycogen. The name myofibrillar myopathy comes from a description of the physical changes we have identified in the muscle cells of affected horses. These changes involve disruption of the orderly alignment of the contractile proteins called myofibrils.
A normal muscle cell that contains an orderly alignment of contractile proteins arranged within myofibrils packaged longitudinally the muscle cell.
A normal portion of a muscle cell. Desmin (yellow) maintains the orderly alignment of myofibrils. In myofibrillar myopathy, the myofibrils form clumps and the orderly alignment of the pink myofrbrils is disrupted.
Source: J Clin Invest. 2009;119(7):1806-1813.
A diagnosis of MFM requires a special microscopic stain for desmin in a muscle biopsy. Equine MFM does not necessarily have the same underlying cause or the same clinical severity as human disorders that are called myofibrillar myopathy. Do not expect to find the same symptoms in horses as described in humans. Human and equine myofibrillar myopathy share the same name because they have a similar, but less severe appearance under the microscope. Often, horses have milder symptoms than humans.
What are the symptoms of MFM in horses?
In Arabians, particularly those competing in endurance, the most common clinical sign of MFM is intermittent tying up (episodes of muscle pain, stiffness, reluctance to move). The severity of muscle stiffness can be much milder than that seen with classic tying-up (exertional rhabdomyolysis). For example, horses may appear slightly stiff and have dark urine without being completely unwilling to move after an endurance ride, or if horses have been rested for a few weeks, marked muscle stiffness often occurs approximately 5 miles into a light ride. Increased serum CK and AST levels can occur but may not be as high as in classic forms of tying-up.
Warmblood horses with MFM may have a history of satisfactory performance as young animals but decline in performance as they reach 8 -10 yrs of age. Tying-up is usually very intermittent or absent in Warmbloods. The most common clinical signs of MFM in Warmbloods are related to poor performance without elevations in serum CK and AST activity. Unwillingness to go forward, a vague, poorly localized hindlimb lameness, sore muscles and drop in energy level are common complaints. Reluctance to collect and engage the hindquarters, poor rounding over fences, poor quality of the canter and slow onset of muscle wasting (atrophy), especially when out of work, are reported. Note that these signs are not specific to MFM and they can occur with poor saddle fit, gastric ulcers, lameness from hock or stifle degeneration, suspensory injuries, sacroiliac disorders, inflammation of thoracic or lumbar vertebrae and more. A thorough evaluation for lameness is needed to rule out these more common issues and should precede any testing for MFM in horses. MFM is much less common than these afore-mentioned equine maladies.
How is a diagnosis of equine MFM established?
A diagnosis of MFM is made by identifying aggregates of a cytoskeletal protein called desmin in a muscle biopsy using immunohistochemical stains. Normally, desmin is found at one specific repeating place in the myofibril, the Z-disc, and it acts to hold myofibrils in proper alignment across the muscle cell. In horses with MFM, sections of a muscle cell start to produce abnormal amounts and shapes of desmin, likely as a reaction to instability of the contractile proteins.
A false positive diagnosis of MFM can occur if the muscle biopsy is taken from a horse with actively regenerating muscle fibers. An experienced evaluator recognizes the difference between young regenerating fibers and mature fibers with desmin aggregates. Additionally, a false negative diagnosis can occur if a horse is too young, the muscle samples are too small. Abnormal desmin staining may not be obvious in horses less than 8 yrs of age, horses out of work for an extended period of time or horses managed with our diet and exercise recommendations. It is possible that younger horses have MFM, but at this point, desmin may not yet have accumulated enough in muscle fibers to identify the disease. Desmin may be a late-stage marker of MFM and we are working to identify more sensitive markers.
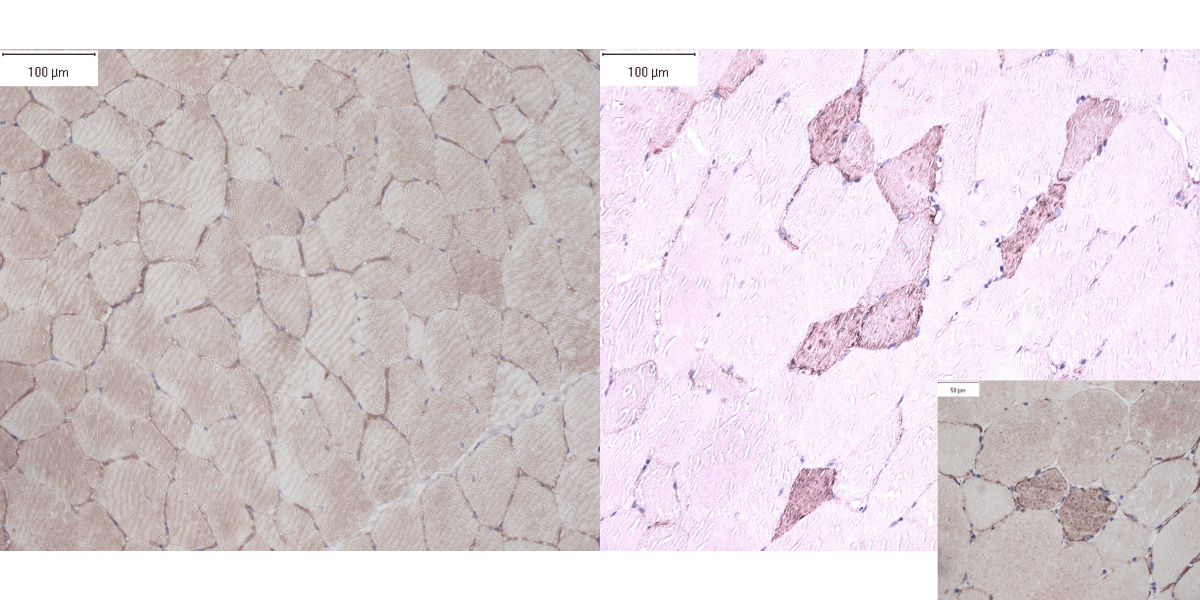
Figure A (Left Side). A normal desmin stain of equine skeletal muscle where a normal amount of desmin is seen under the cell membranes.
Figure B (Right Side). A desmin stain of skeletal muscle from a horse with MFM. Abnormal red clumping of desmin occurs in scattered muscle fibers.
Is PSSM2 the same disease as MFM?
PSSM2 is a term used to describe horses with the microscopic appearance of clumped glycogen in their muscle. Recent research has shown that, in Quarter Horses, PSSM2 is associated with excessive concentrations of glycogen in muscle. In Arabians and Warmbloods previously diagnosed with PSSM2 by microscopy, there are not excessive concentrations of glycogen suggesting a different basis for this disease. Thus, there appear to be several diseases grouped under the term PSSM2. MFM appears to be one of these diseases that has a different cause than PSSM2 in Quarter Horses. We believe pooling of glycogen between the breaks in myofibrils is the reason many cases of MFM were previously diagnosed with PSSM2.
Can a muscle biopsy be used for pre-purchase examinations or breeding decisions?
We do not recommend using a muscle biopsy for breeding decisions or for pre-purchase examinations. The desmin stain used to diagnose MFM cannot predict the future. Absence of desmin staining does not mean the horse will always be healthy. The value of a muscle biopsy is in determining what is happening in the muscle in symptomatic horses at the time of biopsy and what type of therapy could be useful.
Is there a genetic test for equine MFM?
Dr. Valberg is not currently using or recommending the genetic testing for the diagnosis of PSSM2 or MFM as there is currently no scientifically validated evidence that the variants for which genetic testing is available are linked to PSSM2 or MFM. To date (January 2023) we have not found any peer-reviewed studies offered by the company providing the commercial tests.
We have now performed three peer-reviewed studies evaluating the commercial genetic test variants P2, P3 and P4 and the results of those studies have not found the commercial genetic test variants to be consistently present in Warmblood or Arabian horses diagnosed with MFM by desmin staining and consistently absent in healthy horses. Nor are they consistently present in Quarter Horses diagnosed with PSSM2 by glycogen stains.
- Williams et al 2020, Candidate gene expression, coding sequence variants and muscle fiber contractile force in Warmblood horses with myofibrillar myopathy, Equine Vet J, https://doi.org/10.1111/evj.13.
- Valberg et al 2020 Commercial genetic testing for type 2 polysaccharide storage myopathy and myofibrillar myopathy does not correspond to a histopathologic diagnosis. Equine Vet J. https://doi.org/10.1111/evj.13.
- Valberg SJ, Williams ZJ, Finno CJ, Schultz A, Velez‐Irizarry D, Henry ML, Gardner K, Petersen JL. Type 2 polysaccharide storage myopathy in Quarter Horses is a novel glycogen storage disease causing exertional rhabdomyolysis. Equine Veterinary Journal. 2022 Aug 16. https://doi.org/10.1111/evj.13876.
A summary of our peer reviewed studies:
Researchers from Michigan State University, University of California-Davis, University of Nebraska-Lincoln and Oregon State University evaluated how often the P2, P3 and P4 variants occur in Warmblood and Arabian horses diagnosed with myofibrillar myopathy (MFM). The variants in MYOT (P2), FLNC (P3) and MYOZ3 (P4) used for commercial tests (genotype) were analyzed in the researchers’ laboratory. We determined how often the variants occurred in 30 healthy endurance Arabians and 54 Warmbloods with no abnormalities in their muscle biopsies compared to 30 endurance Arabians and 54 Warmbloods diagnosed with either PSSM2 or MFM based on changes in their muscle biopsies. The changes we evaluated in the muscle biopsies were the changes that were used to discover these diseases. We found no statistical association between any of the P test variants, or combinations of P test variants, and the presence of either PSSM2 or MFM considered as one diagnosis, or when PSSM2 or MFM were considered separately. Healthy control horses were as likely to possess a P2, P3 or P4 variant as a horse with PSSM2 or MFM. By chance alone, 29% of Warmbloods and 25% of Arabians will have at least one P variant regardless of whether they have a muscle disease or not. We also looked at how commonly the P variants occurred in publicly available genetic databases of 205 horses of a wide variety of breeds. We found that the P variants appear to be common genetic variants in horses of many different breeds. Thus, we found no evidence to support that the P variants are themselves causative or diagnostic of a muscle disease in the horse. The use of the P variant genetic tests in selection decisions, pre-purchase examination or diagnosis of a myopathy was not supported by the findings in our two research studies.
It is important to be aware that when comparing the genome of one healthy horse to another, there are as many as 17 million genetic variants that make an individual unique. In other words, it is common to find genetic variants between horses, even in important and highly functional genes. It is also common to find genetic variants that are shared between horses. The difficulty with genetic disease research is not in finding variants, but rather is determining whether a specific variant is responsible for a specific disease. Scientifically validated variants such as those used in the AQHA 6 panel genetic test have undergone peer review as part of the validation process and are published in peer-reviewed publications.
Peer review and publication is the foundation of sound scientific research. Marketing tests as providing conclusive answers to complex genetic diseases before scientific peer review may result in misdiagnosis with adverse consequences. For more information about the pitfalls of poorly validated genetic testing, please reference the article “Pet genomics medicine runs wild” in the prestigious journal, Nature.
To see an example of how scientific validation works, see the recent open access publication on the genetic basis for immune-mediated myositis (IMM) in horses.
What breeds are diagnosed with MFM?
The majority of horses we have diagnosed with MFM have been Arabian and Warmblood horses or their crosses. Very rarely do we find MFM in Thoroughbreds, Quarter Horses and Paso diagnosed by muscle biopsy. We do not have an estimate of how common MFM is in equine breeds.
What causes MFM?
MFM appears to be a complex trait, meaning that environmental influences and expression of many genes play a role in the development of disease. Based on our research, we believe it is unlikely one specific gene causes MFM. Based on gene and protein expression studies of Arabian horses, MFM in Arabians appears to involve altered cysteine metabolism. Cysteine is an amino acid that is a key structural component of proteins and a necessary component of many antioxidants in muscle. Endurance horses have a high capacity to oxidize fatty acids which can create oxidative stress during aerobic exercise. A deficiency of cysteine-containing antioxidants could cause chronic oxidation and aggregation of key proteins such as desmin.
During normal muscle contractions, signaling pathways in muscle fibers detect the magnitude and dynamics of muscle contractions thus fine-tuning cellular adaptations in protein production to strengthen the fiber and enhance energy generation. Results of gene and protein expression studies suggest that Warmblood MFM arises from abnormal adjustment of signaling pathways to the demands of exercise, resulting in a lack of muscle repair, damage to mitochondrial components and oxidative stress. Disease expression could be due to the combined impact of the magnitude of exercise-induced mechanical forces, dietary interactions and aberrant mechano-signaling, but this needs further evaluation.
How do I manage a horse with MFM?
First, obtain a specific diagnosis from a qualified individual. If horses have signs of exercise intolerance, we strongly recommend organizing a complete veterinary examination. Many different types of lameness can cause clinical signs similar to MFM, thus a thorough evaluation is needed before a muscle biopsy is submitted or changes to diet and exercise are implemented.
Where can I find out more about MFM?
Please do NOT use the human literature as a guide to understanding equine MFM, the clinical picture IN HUMANS does not correlate well with horses.
Current research publications include:
- Valberg SJ, McKenzie EC, Eyrich LV, Shivers J, Barnes N, Finno CJ. Suspected myofibrillar myopathy in Arabian horses with a history of exertional rhabdomyolysis. Equine Vet J. 2016:48;548–556.
- McKenzie EC, Eyricha ME, Valberg SJ. Clinical, histopathological and metabolic responses following exercise in Arabian Horses with a history of exertional rhabdomyolysis. Vet J. 2016 Oct;216:196-201.
- Valberg SJ, Nicholson AM, Lewis SS, Reardon RA, C.J. Finno. Clinical and histopathological features of myofibrillar myopathy in Warmblood horses. Equine Vet J. 2017 Nov;49(6):739-745.
- Valberg SJ, Perumbakkam S, McKenzie EC, Finno CJ. Proteome and transcriptome profiling of equine myofibrillar myopathy identifies diminished peroxiredoxin 6 and altered cysteine metabolic pathways. Physiol Genomics. 2018 Dec 1;50(12):1036-1050. doi: 10.1152/physiolgenomics.00044.2018. Epub 2018 Oct 5.
- Williams et al 2020, Candidate gene expression, coding sequence variants and muscle fiber contractile force in Warmblood horses with myofibrillar myopathy, Equine Vet J, https://doi.org/10.1111/evj.13.
- Valberg SJ, Finno CJ, Henry ML, Schott M. Petersen JL Commercial genetic testing for type 2 polysaccharide storage myopathy and myofibrillar myopathy does not correspond to a histopathologic diagnosis. Equine Vet J 2020, https://doi.org/10.1111/evj.13.
- Williams ZJ, Velez-Irizarry D, Gardner K, Valberg SJ. Integrated proteomic and transcriptomic profiling identifies aberrant gene and protein expression in the sarcomere, mitochondrial complex I, and the extracellular matrix in Warmblood horses with myofibrillar myopathy. BMC genomics. 2021 Dec;22(1):1-20. https://doi.org/10.1186/s12864-021-07758-0.